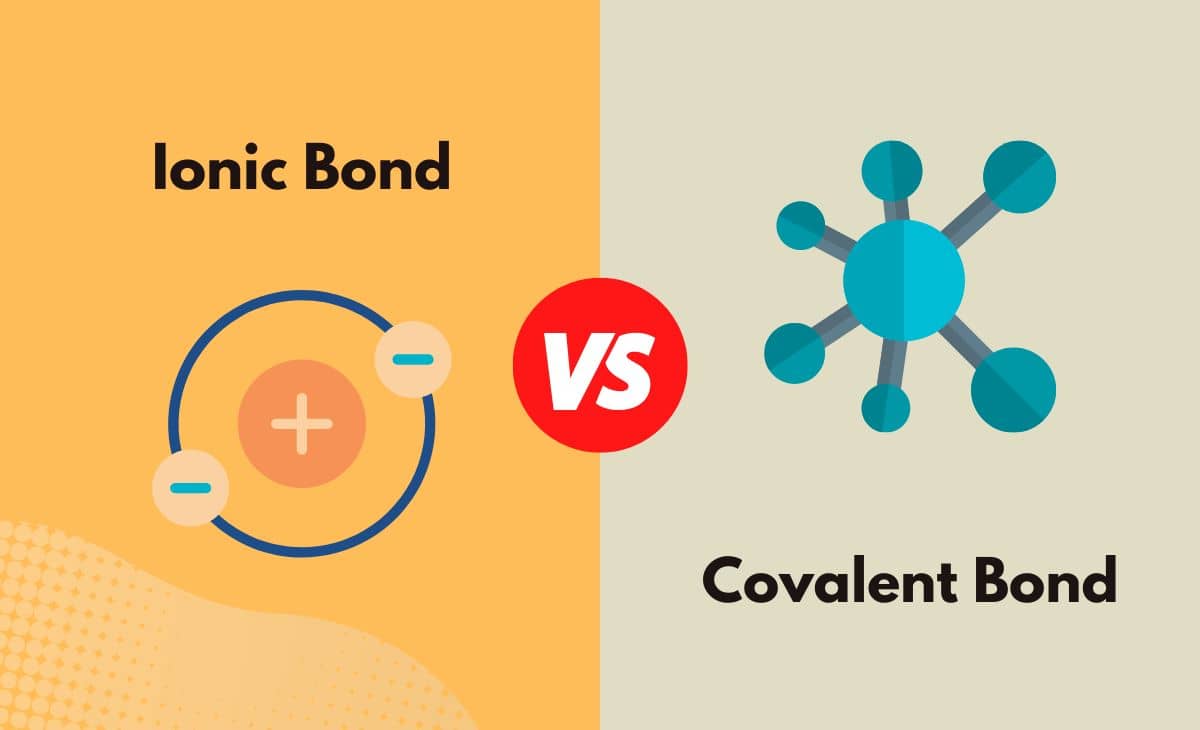
Covalent bonding is the stable balance of attractive and repulsive forces between two atoms as they share the electron. In a covalent bond the two atoms come together to share the electron, instead of an atom taking an electron from another. When they are in water, they also have a high electrical conductivity.Ī covalent bond is another strong chemical bond. They are highly soluble in water and have a high melting point. Ionic compounds are usually in the form of solids, i.e. As opposites attract, these ions are then strongly attracted to each other, so strongly that they become bonded. The non-metal ion which gained an electron has a negative charge. The metal ion which lost an electron has a positive charge. Hence both atoms become ions as one atom has one electron less, while the other has an extra. a metal and a non-metal) atoms in which one atom gives up an electron to another. The two common examples of ‘strong bonds’ are the ionic bonds and covalent bonds.Īn ionic bond is a chemical bond between two dissimilar (i.e. The strength of these chemical bonds can be loosely classified as ‘strong bonds’ or ‘weak bonds’. The chemical bond is in fact caused by the electrostatic force of attraction between opposite charges, either between electrons and nuclei, or as the result of a dipole attraction. A bond is a chemical process born from an attraction between two or more atoms. In order to create molecules or compounds, atoms form bonds with each other. A covalent bond is another strong chemical bond. Key difference: An ionic bond is a chemical bond between two dissimilar (i.e.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
